Thermodynamics: Heat
- Mole
- Gas Volume
- States of Matter
- Heat 🢀
- Enthalpy
- Thermodynamics
- Adiabatic Process
- Mass Energy Conservation
- Carnot Engine
Heat, Temperature and Internal Energy
Heat, like work, represents a transfer of energy. Heat is defined as a transfer of energy due to a difference of temperature. When two objects at different temperatures are put in contact, heat spontaneously flows from the hotter one to the colder one until both objects have the same temperatures. The SI unit of heat 1 joule (J). An often used unit is 1 calorie (cal) which is the heat needed to raise the temperature of 1 g of water (at 15°C) by 1°C:
1 cal = 4.186 J
1 kcal = 4.186 kJ
Temperature is a measure of the average kinetic energy of individual molecules within an object. It is independent of the mass of the object (so called intensive property). Internal energy refers to the total energy of all the molecules. It grows with the mass (extensive property). The relation between the temperature T (in Kelvin) and the the internal energy U is (for ideal monatomic gas):
2
Specific heat
The heat Q required to change the temperature of a given material is proportional to the mass m of the material present and to the temperature change ΔT:
The proportionality constant c, which depends on the material, is called the specific heat. If ΔT > 0 the heat flows into the object, if ΔT < 0 the heat flows out of the object.
Adding heat to gases not only raises their temperature but can increase also their volume and pressure. According to the First Law of thermodynamics we have:
where W is the work done by the gas which occurs when the gas expands. We distinguish:
- cp: specific heat at constant pressure (or isobaric). This constant is used for all processes (e.g. adiabatic) where the volume changes and the gas does work Enthalpy.
- cv: specific heat at constant volume (or isochoric). Since the gas does not generate any work all added heat goes into the increased temperature, so cv < cp.
When we put several objects at different temperatures Ti together the overall system will approach one temperature T that can be calculated from the equation of the energy conservation:
For gases the specific heats are different for constant pressure cp and for constant volume cv processes.
Latent heat
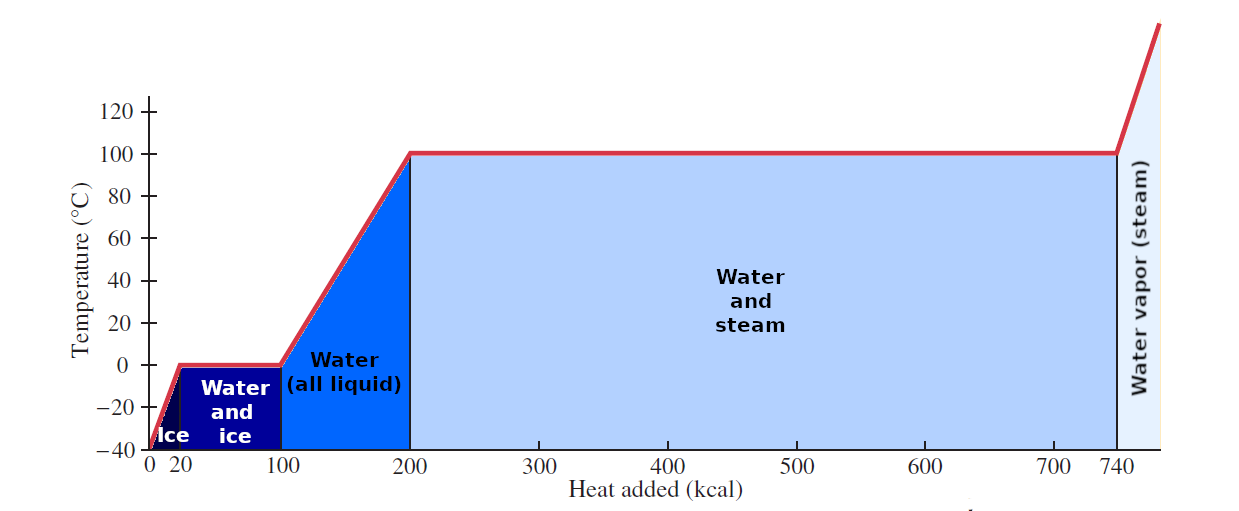
When we warm ice its temperature rises until it reaches 0°C and it begins melting. Further addition of heat does not rise the temperature of the ice/liquid mixture until all ice has melt. An anologuos situation occurs when heat is added to boiling water at 100°C. The heat required to change 1.0 kg of a substance from the solid to the liquid state is called the latent heat of fusion LF, for ice it is LF = 80 kcal/kg = 333 kJ/kg. The heat required to change 1.0 kg of a substance from the liquid to the vapor (gas) state is called the latent heat of vaporization LV, for water it is LV = 540 kcal/kg = 2260 kJ/kg. The heats of vaporization and fusion also refer to the amount of heat released by a substance when it changes from a gas to a liquid, or from a liquid to a solid. The heat involved in a change of phase for the mass m of a substance is
Heat Transfer: Conduction
Heat is transferred from one place or object to another in three different ways: by conduction, by convection, and by radiation.
Heat conduction is heat transfer through materials caused by molecular collisions. In 1-dimensional case we have for the heat flow:
dt
dx
where
| Q/dt | heat flow per second |
| k | thermal conductivity which is characteristic of the material |
| A | area through which the heat flows |
| dT/dx | temperature gradient over the distance dx. This gradient is negative because T(x+dx) < T(x) (heat flows from higher to lower temperature) |
The insulating properties of building materials are often specified by R-values (or "thermal resistance"), defined for a given thickness dx of material as R = dx / k. Good insulators have a high value of R.
Heat Transfer: Convection
Heat convection is heat transfer caused by moving substances carrying thermal energy. Examples: hot air rise, air movement due to a ventilator, Gulf stream, etc.
Heat Transfer: Radiation
Radiation consists essentially of electromagnetic waves, so it does not require the presence of matter as a medium to carry the heat. The rate at which an object radiates energy is proportional to the area A of the emitting object and to the fourth power of the Kelvin temperature, A:
dt
Eq. (6) is called the Stefan-Boltzmann equation, and σ is a universal constant called the Stefan-Boltzmann constant. The factor ε called the emissivity, is a number between 0 and 1 that is characteristic of the surface of the radiating material. Very black surfaces have emissivity close to 1, whereas shiny metal surfaces have close ε to zero.
Objects not only emit radiation, they also absorb radiation according to Eq. (6) with the same constants ε, σ but a different temperature. If an object has temperature T1 and its surroundings has temperature T2 the net rate of radiant heat flow from the object is
dt
Heating of an object by radiation from the Sun cannot be calculated using Eq. (7) since this equation assumes a uniform temperature T2, of the environment surrounding the object, whereas the Sun is essentially a point source.